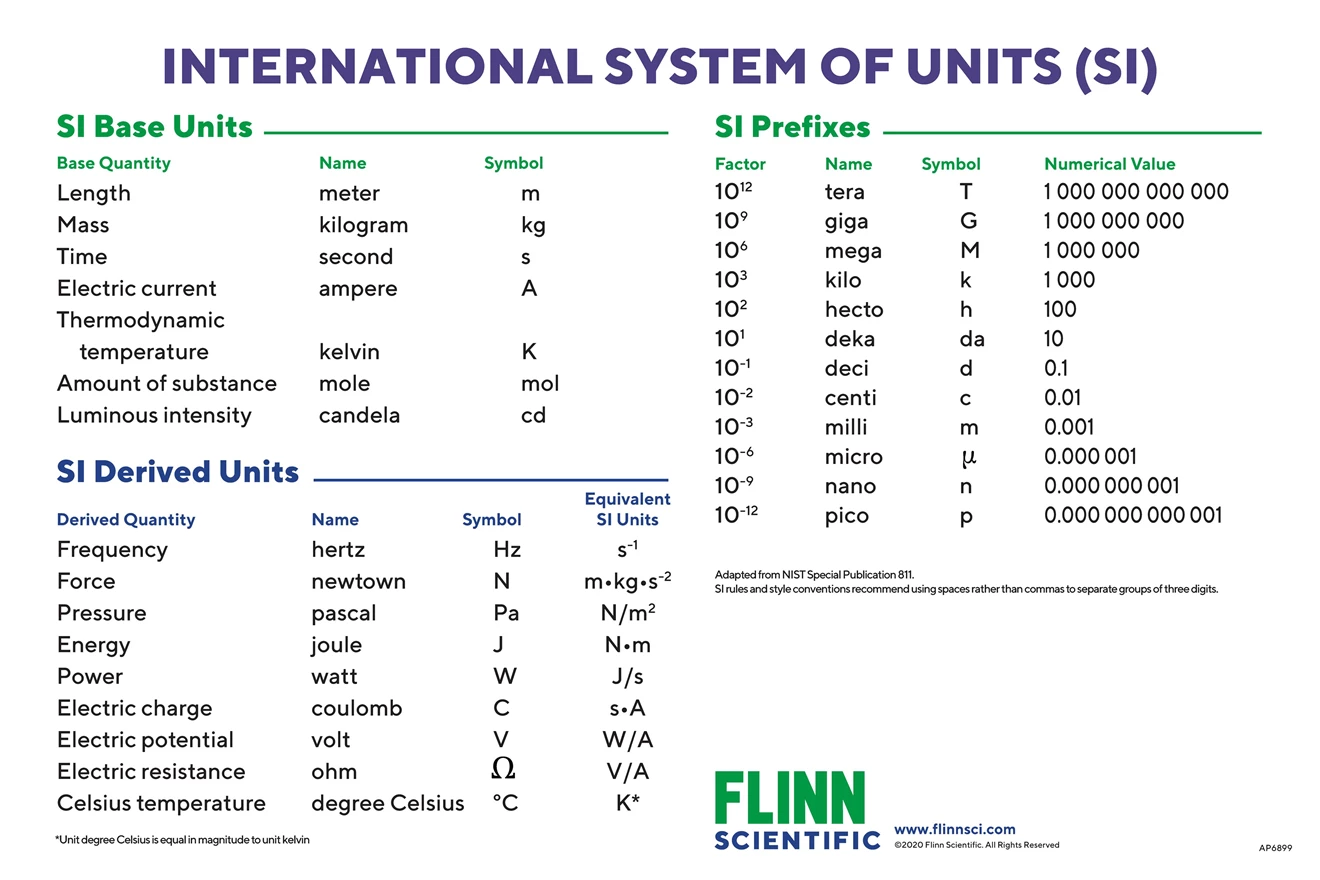
Chem Notes Mole- SI unit for amount of matter Mole- SI unit for amount of matter mol 6.02 X representative particles= Avogadro's Number. - ppt download

Chapter 7 Chemical Quantities. The Mole SI base unit to measure the amount of a substance 1 mole of anything = x representative particles. - ppt download

A new definition of the mole has arrived - IUPAC | International Union of Pure and Applied Chemistry